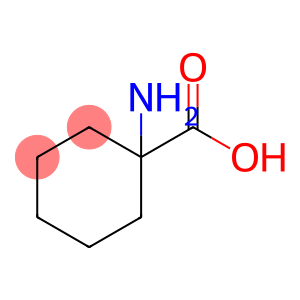
1-aminocyclohexanecarboxylic acid
1-Amino-1-cyclohexanecarboxylic acid
CAS: 2756-85-6
Molecular Formula: C7H13NO2
1-aminocyclohexanecarboxylic acid - Names and Identifiers
| Name | 1-Amino-1-cyclohexanecarboxylic acid |
| Synonyms | H-AC6C-OH HOMOCYCLOLEUCINE IFLAB-BB F3095-1803 RARECHEM AK HZ 0008 LABOTEST-BB LT01138435 1-Aminocyclohexanecarboxylic acid 1-aminocyclohexanecarboxylic acid 1-Amino-cyclohexanecarboxylic acid 1-ammonio-1-cyclohexanecarboxylate 4-aminopiperidine-4-carboxylic acid 1-Amino-1-cyclohexanecarboxylic acid 1-AMINOCYCLOHEXANECARBOXYLIC ACID-HBr 1-Amino-1-cyclohexane carboxylic acid |
| CAS | 2756-85-6 |
| EINECS | 220-411-0 |
| InChI | InChI=1/C6H12N2O2/c7-6(5(9)10)1-3-8-4-2-6/h8H,1-4,7H2,(H,9,10) |
| InChIKey | WOXWUZCRWJWTRT-UHFFFAOYSA-N |
1-aminocyclohexanecarboxylic acid - Physico-chemical Properties
| Molecular Formula | C7H13NO2 |
| Molar Mass | 143.18 |
| Density | 1.1095 (rough estimate) |
| Melting Point | >300°C(lit.) |
| Boling Point | 261.28°C (rough estimate) |
| Flash Point | 125.4°C |
| Water Solubility | almost transparency |
| Vapor Presure | 0.000812mmHg at 25°C |
| Appearance | Crystalline Powder |
| Color | White to almost white |
| BRN | 2355692 |
| pKa | pK1: 2.65(+1);pK2: 10.03(0) (25°C) |
| Storage Condition | Keep in dark place,Inert atmosphere,Room temperature |
| Sensitive | Hygroscopic |
| Refractive Index | 1.4150 (estimate) |
| MDL | MFCD00001487 |
1-aminocyclohexanecarboxylic acid - Risk and Safety
| Hazard Symbols | Xi - Irritant |
| Risk Codes | 36/37/38 - Irritating to eyes, respiratory system and skin. |
| Safety Description | S22 - Do not breathe dust. S24/25 - Avoid contact with skin and eyes. S37/39 - Wear suitable gloves and eye/face protection S26 - In case of contact with eyes, rinse immediately with plenty of water and seek medical advice. |
| WGK Germany | 3 |
| RTECS | GU8393000 |
| FLUKA BRAND F CODES | 3 |
| TSCA | Yes |
| HS Code | 29224999 |
1-aminocyclohexanecarboxylic acid - Reference Information
| EPA chemical information | Information provided by: ofmpub.epa.gov (external link) |
| Use | 1-amino-1-cyclohexylformic acid is a cyclic amino acid. Cyclic amino acids are a configuration-limiting amino acids. Their conformational restrictive characteristics are conducive to the formation and stability of the bioactive conformation of peptides. The conformational effects and biological effects caused by modifying peptides have important theoretical and application values. They are often used to modify bioactive peptides to improve their pharmaceutical properties. Studies have shown that the introduction of cyclic amino acids is one of the effective methods to improve the properties of polypeptides. |
| preparation | cyclohexanone (14.8g,150mmol),NH4CI(8.3g,165mmol), ammonia (20mI,270mmol), absolute ethanol (45mI) and water (30mI) were added to a 250mL round bottom flask. Stir to clarify at room temperature, continue to stir for 3 hours, the solution turns light red. Add NaCN(8.lg,165mmol), airtight. At room temperature, the reaction liquid changes from light red to reddish brown, and a large number of white solids are generated. Distillate under reduced pressure to remove most of the ethanol and ammonia, cool to room temperature, extract with dichloromethane (3 × 30mL), combine organic layers, wash with 2 × lOmL of water, and dry with anhydrous sodium sulfate. Filtration, recovery of dichloromethane, red brown 1-aminocyclohexanonitrile crude 18.0g, 96.8% yield. Directly for the next reaction. Under the ice bath, pre-cooled concentrated hydrochloric acid (75mL) is added to the 1-aminocyclohexylnitrile prepared above, a large amount of heat is released, stirred overnight at room temperature, the solid is dissolved, refluxed for 3 hours, a large amount of solid is generated, distilled to dry under reduced pressure, then 30mL of water is added, and evaporated to dry to obtain yellow residue. Add 15ml. Water, ice bath with 4moI/LNaOH solution adjusted to pH 3~4, stirring overnight at room temperature, filtering, absolute ethanol washing, white solid. The white crystal 12.5g of the target compound was obtained by recrystallization with 75% ethanol aqueous solution in 58.3% yield. |
Last Update:2024-04-09 21:01:54
Supplier List
Spot supply
Product Name: 1-Amino-1-cyclohexanecarboxylic acid Visit Supplier Webpage Request for quotationCAS: 2756-85-6
Tel:
Email: qianyanbiochem@gmail.com
Mobile: 13247110337
QQ: 2972965813
Product List: View Catalog
Spot supply
Product Name: 1-Amino-1-cyclohexanecarboxylic acid Visit Supplier Webpage Request for quotationCAS: 2756-85-6
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 1-Amino-1-cyclohexanecarboxylic acid Request for quotation
CAS: 2756-85-6
Tel: +86 17733984678
Email: alice@jiuzhou-chem.com
QQ: 2284527519
WhatsApp: +86 17733984678
Linkedin: http://postmaster@api-made.com/
CAS: 2756-85-6
Tel: +86 17733984678
Email: alice@jiuzhou-chem.com
QQ: 2284527519
WhatsApp: +86 17733984678
Linkedin: http://postmaster@api-made.com/
Product Name: 1-Aminocyclohexanecarboxylic acid Visit Supplier Webpage Request for quotation
CAS: 2756-85-6
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 2756-85-6
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Spot supply
Product Name: 1-Amino-1-cyclohexanecarboxylic acid Visit Supplier Webpage Request for quotationCAS: 2756-85-6
Tel:
Email: qianyanbiochem@gmail.com
Mobile: 13247110337
QQ: 2972965813
Product List: View Catalog
Spot supply
Product Name: 1-Amino-1-cyclohexanecarboxylic acid Visit Supplier Webpage Request for quotationCAS: 2756-85-6
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 1-Amino-1-cyclohexanecarboxylic acid Request for quotation
CAS: 2756-85-6
Tel: +86 17733984678
Email: alice@jiuzhou-chem.com
QQ: 2284527519
WhatsApp: +86 17733984678
Linkedin: http://postmaster@api-made.com/
CAS: 2756-85-6
Tel: +86 17733984678
Email: alice@jiuzhou-chem.com
QQ: 2284527519
WhatsApp: +86 17733984678
Linkedin: http://postmaster@api-made.com/
Product Name: 1-Aminocyclohexanecarboxylic acid Visit Supplier Webpage Request for quotation
CAS: 2756-85-6
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 2756-85-6
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History



